at 25 degree celsius the molar conductance at infinite dilution for HCl solution is 4.25*10 2 ohm m2 mol , while its specific conductanceis 382.5 ohm m . if degree of dissociation is 90
✓ Solved: Calculate the pH after 0.020 mole of HCl is added to 1.00 L of each of the four solutions in...

A piece of metallic iron (10 moles) was dissolved in concentrated hydrochloric acid. The reaction formed hydrogen gas and iron chloride. How many grams of HCl were consumed? Don't forget the units. {
Assuming that: Part I (molarity of HCl by standardization): Part II (determination of sodium carbonate in unknown) Solution: Mas
Hydrochloric acid, 500 ml, glass | Reagents for Decalcification | Reagents for Histology | Histology/Microscopy | Life Science | Carl Roth - International

10 moles of HCL is added to excess to magnesium and forms 4 moles of hydrogen gas then Percentage - Brainly.in

SOLVED: 1 How many moles of HCl are there in 10 mL of a solution with a concentration of 0.5 molL-I? swered Select one: 5 mole 0.05 mole of 200 Jestion 50 mole 0.5 mole
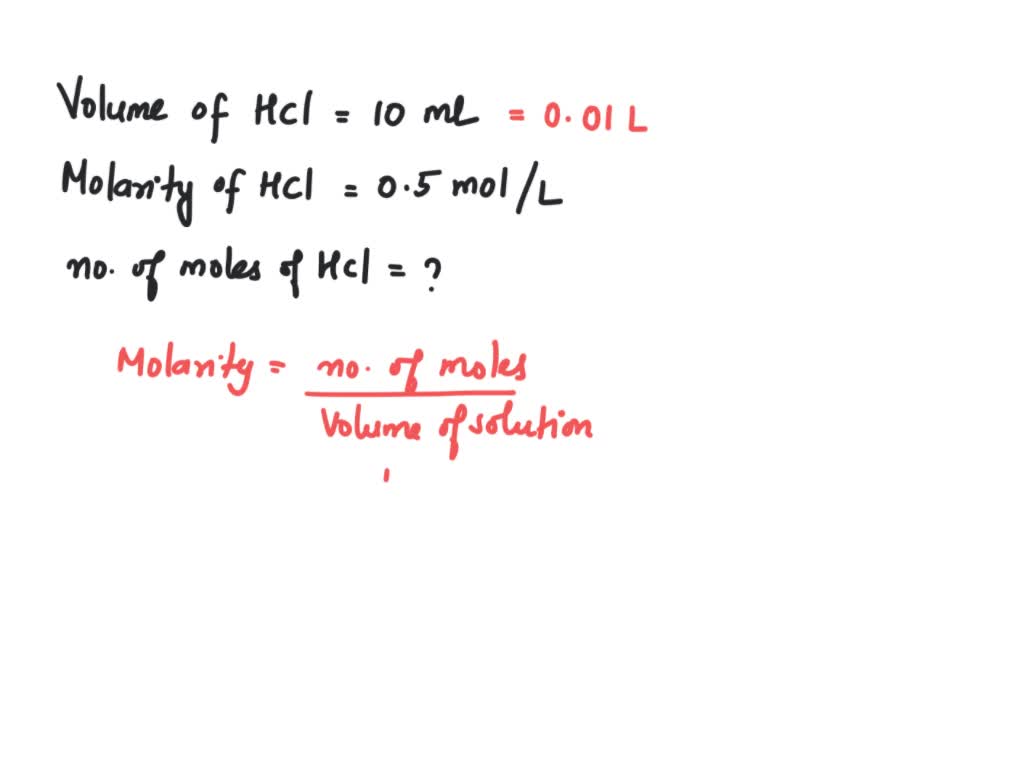
SOLVED: 1 How many moles of HCl are there in 10 mL of a solution with a concentration of 0.5 molL-I? swered Select one: 5 mole 0.05 mole of 200 Jestion 50 mole 0.5 mole




![ANSWERED] A flask contains 0.015 mol of HCl in 10.... - Inorganic Chemistry ANSWERED] A flask contains 0.015 mol of HCl in 10.... - Inorganic Chemistry](https://media.kunduz.com/media/sug-question/raw/55747522-1659637855.5533836.jpeg)