If `NaCl` is doped with `10^(-3)` mol% of `SrCl_(2)`, what is the concentration of cation vacanc... - YouTube

If Avogadro number NA is changed from 6.022 × 10^23 mol^-1 to 6.022 × 10^20 mol^-1 , this would change :

Main-chain engineering of polymer photocatalysts with hydrophilic non-conjugated segments for visible-light-driven hydrogen evolution | Nature Communications

Oxidation of secondary alcohols using 10 mol% lanthanum triflate as... | Download Scientific Diagram

Catalysts | Free Full-Text | Organocatalyzed Michael Addition to Nitroalkenes via Masked Acetaldehyde
For a reaction, N2(g)+3H2(g) →2NH3 (g), the rate of formation of NH_3 was found to be 2 x 10^ 2 mol/dm3/s. The rate of consumption of N2 will b
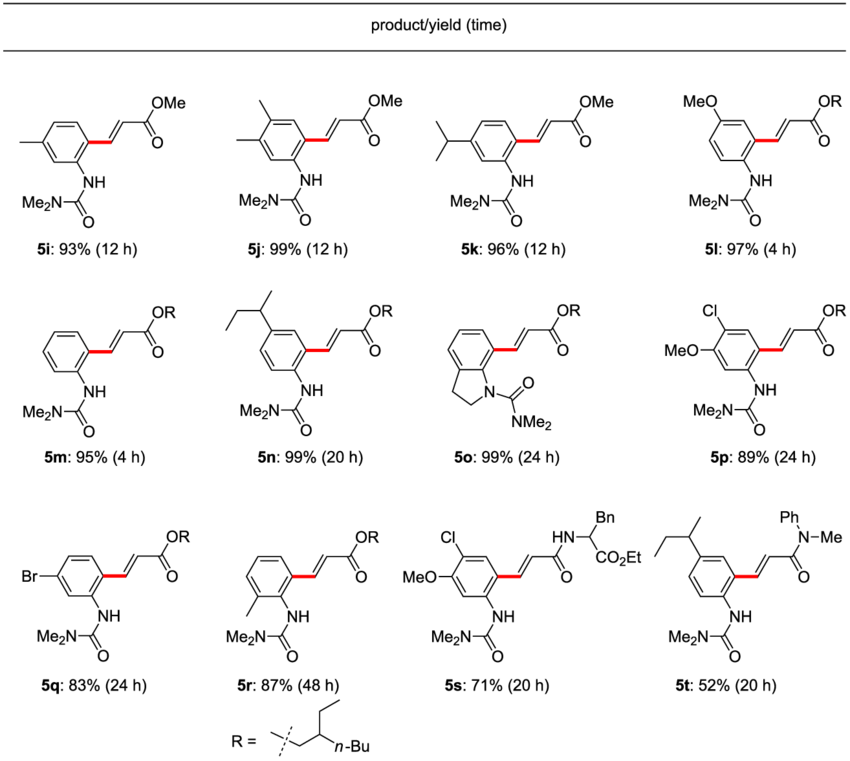
Representative results in EtOAc. Conducted at rt in EtOAc with 10 mol %... | Download Scientific Diagram
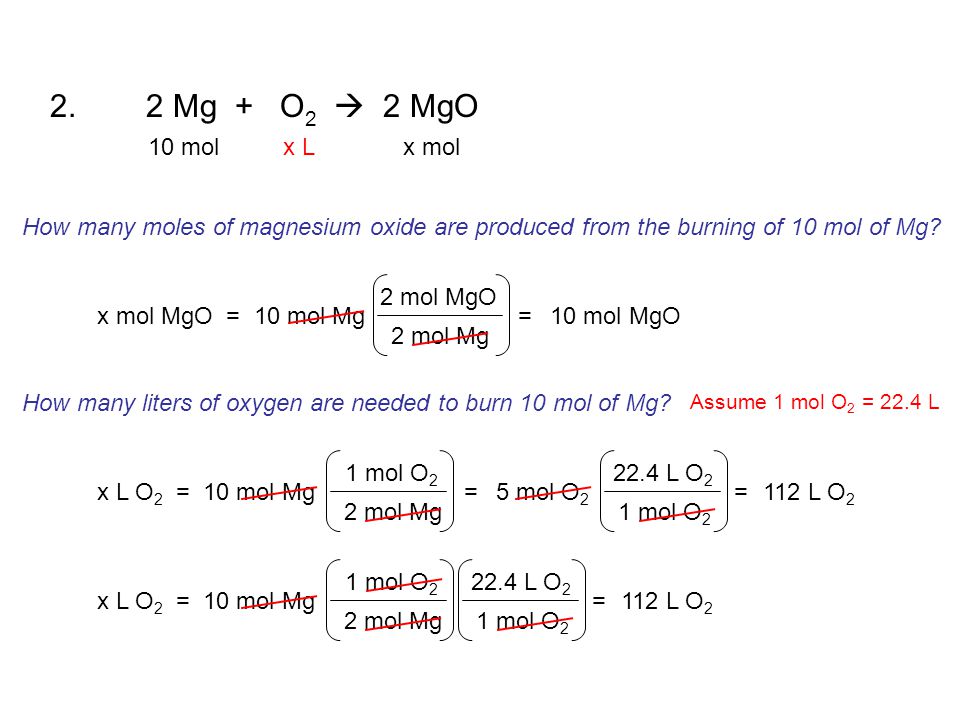
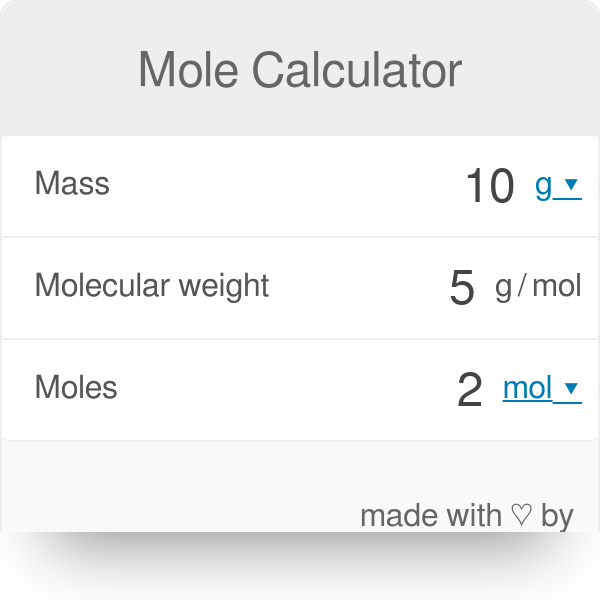
Mole Calculations. ? Visualizing a Chemical Reaction Na + Cl 2 NaCl ___ mole Cl 2 ___ mole NaCl___ mole Na ppt download