For complete combustion of ethanol . C2H5OH(l)+3O2(g)=2CO2(g)+3H2O(l) the amount of heat produced as measured in bomb calorimeter ,is 1364.47kJ/mol at 25degree celsius . Assuming ideality the enthalpy of combustion ,DelHc For the

Dihydrogen gas is obtained from natural gas by partial oxidation with steam as per following endothermic reaction: CH4 (g) + H2O (g) CO (g) + 3H2 (g) (a) Write as expression for
![Ground and Excited States of the [Fe(H2O)6]2+ and [Fe(H2O)6]3+ Clusters: Insight into the Electronic Structure of the [Fe(H2O)6]2+–[Fe(H2O)6]3+ Complex | Journal of Chemical Theory and Computation Ground and Excited States of the [Fe(H2O)6]2+ and [Fe(H2O)6]3+ Clusters: Insight into the Electronic Structure of the [Fe(H2O)6]2+–[Fe(H2O)6]3+ Complex | Journal of Chemical Theory and Computation](https://pubs.acs.org/cms/10.1021/ct501143c/asset/images/large/ct-2014-01143c_0017.jpeg)

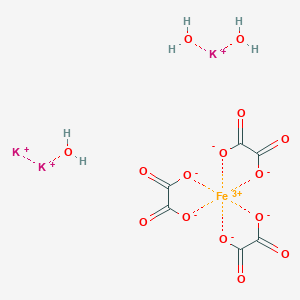


![Warum ist [Fe(H2O)6]3+ farblos und [Fe(H2O)3(SCN)3] tiefrot? - ppt video online herunterladen Warum ist [Fe(H2O)6]3+ farblos und [Fe(H2O)3(SCN)3] tiefrot? - ppt video online herunterladen](https://slideplayer.org/901912/3/images/slide_1.jpg)


![H2O: Just Add Water The Complete Season 3 [4 Discs] [DVD] - Best Buy H2O: Just Add Water The Complete Season 3 [4 Discs] [DVD] - Best Buy](https://pisces.bbystatic.com/image2/BestBuy_US/images/products/2093/20938694_sa.jpg)







