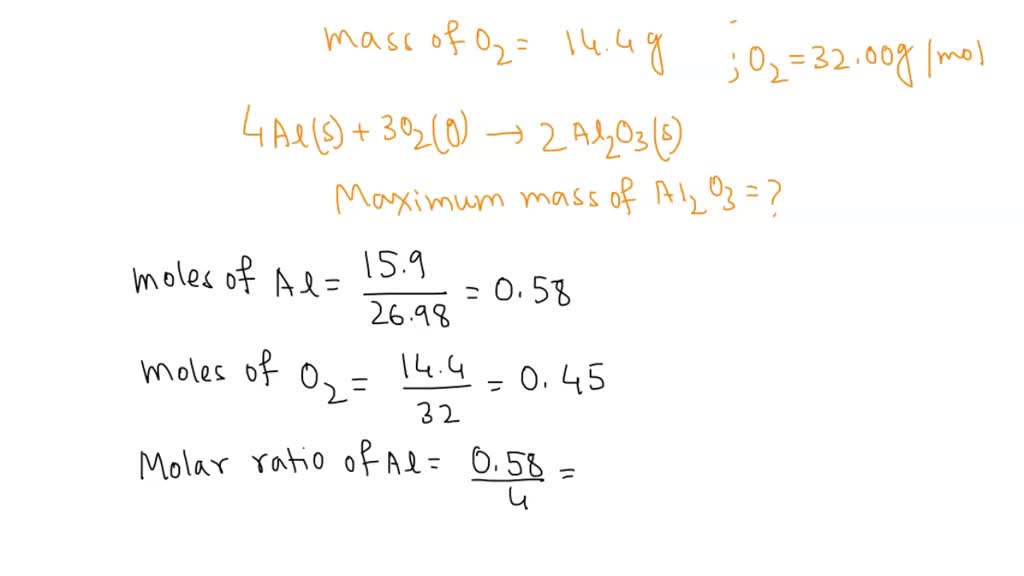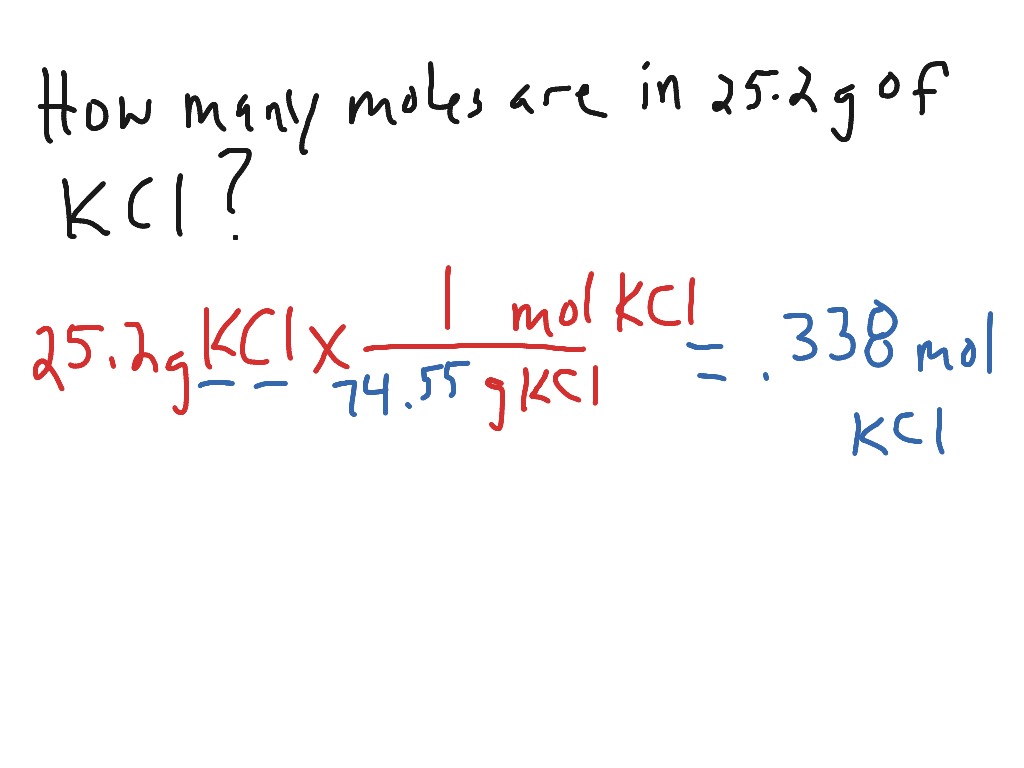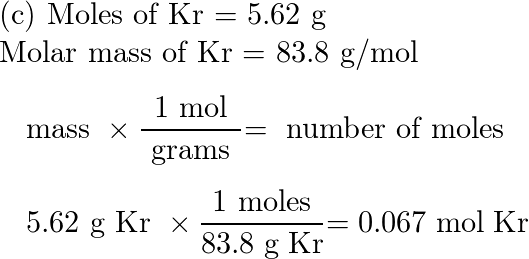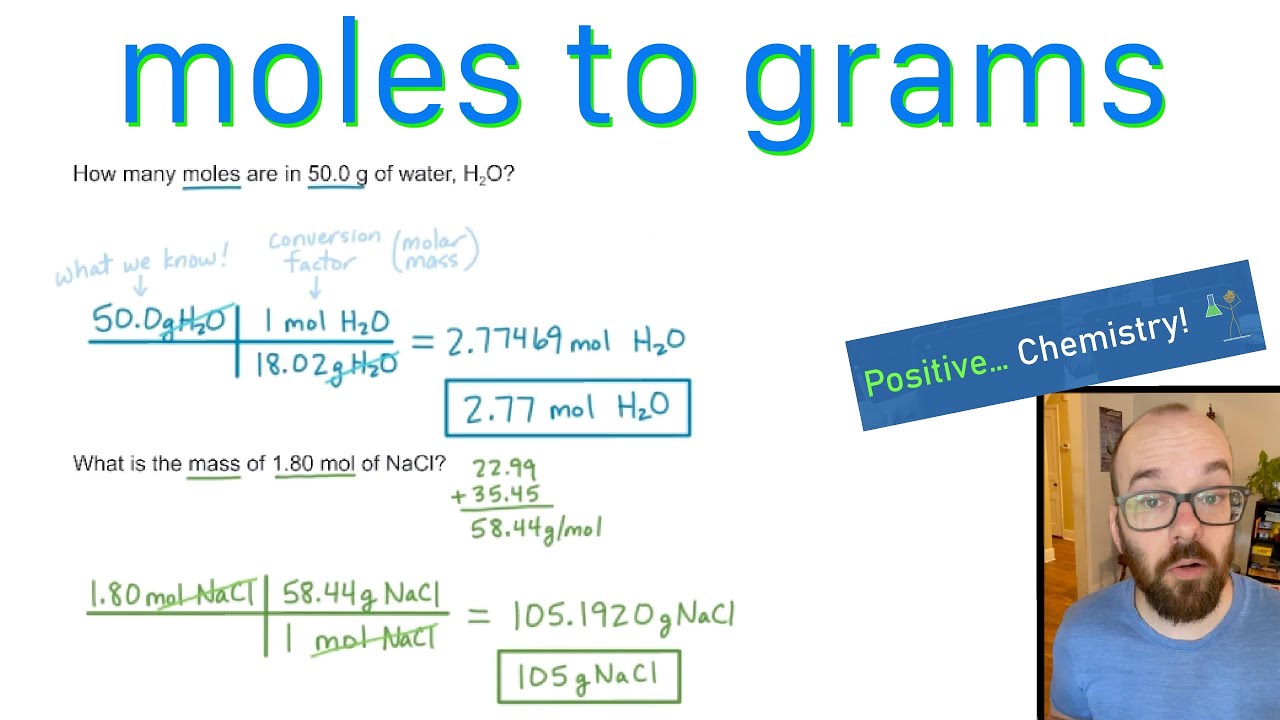Analysis of Two Definitions of the Mole That Are in Simultaneous Use, and Their Surprising Consequences | Journal of Chemical Education

Answered: 54.0g Al reacts with 64.0g O2 to form Al2O3 according to the equation. 4Al+3O2 = 2Al2O3 O2: 32 - Brainly.com