![SOLVED: Which of the statements outlined below is NOI correct regarding the relationship between the hydronium and hydroxide ion concentrations, [H3O*] and [OH ], respectively, in relation to the pH of an SOLVED: Which of the statements outlined below is NOI correct regarding the relationship between the hydronium and hydroxide ion concentrations, [H3O*] and [OH ], respectively, in relation to the pH of an](https://cdn.numerade.com/ask_images/2c1d38b061434304a23e9db2b01d94f0.jpg)
SOLVED: Which of the statements outlined below is NOI correct regarding the relationship between the hydronium and hydroxide ion concentrations, [H3O*] and [OH ], respectively, in relation to the pH of an

The Nature of the H3O+ Hydronium Ion in Benzene and Chlorinated Hydrocarbon Solvents. Conditions of Existence and Reinterpretation of Infrared Data | Journal of the American Chemical Society

![Answered: Calculate the concentration of [H3O+]… | bartleby Answered: Calculate the concentration of [H3O+]… | bartleby](https://content.bartleby.com/qna-images/question/735b0976-906f-40ec-a76b-1f1d9f7b453e/2596ed75-d4c3-4ac0-b3bc-7ff8d10682b8/6e0bvvf_processed.jpeg)
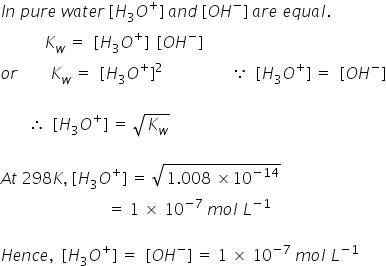
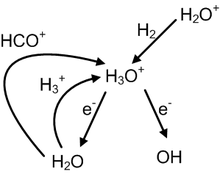


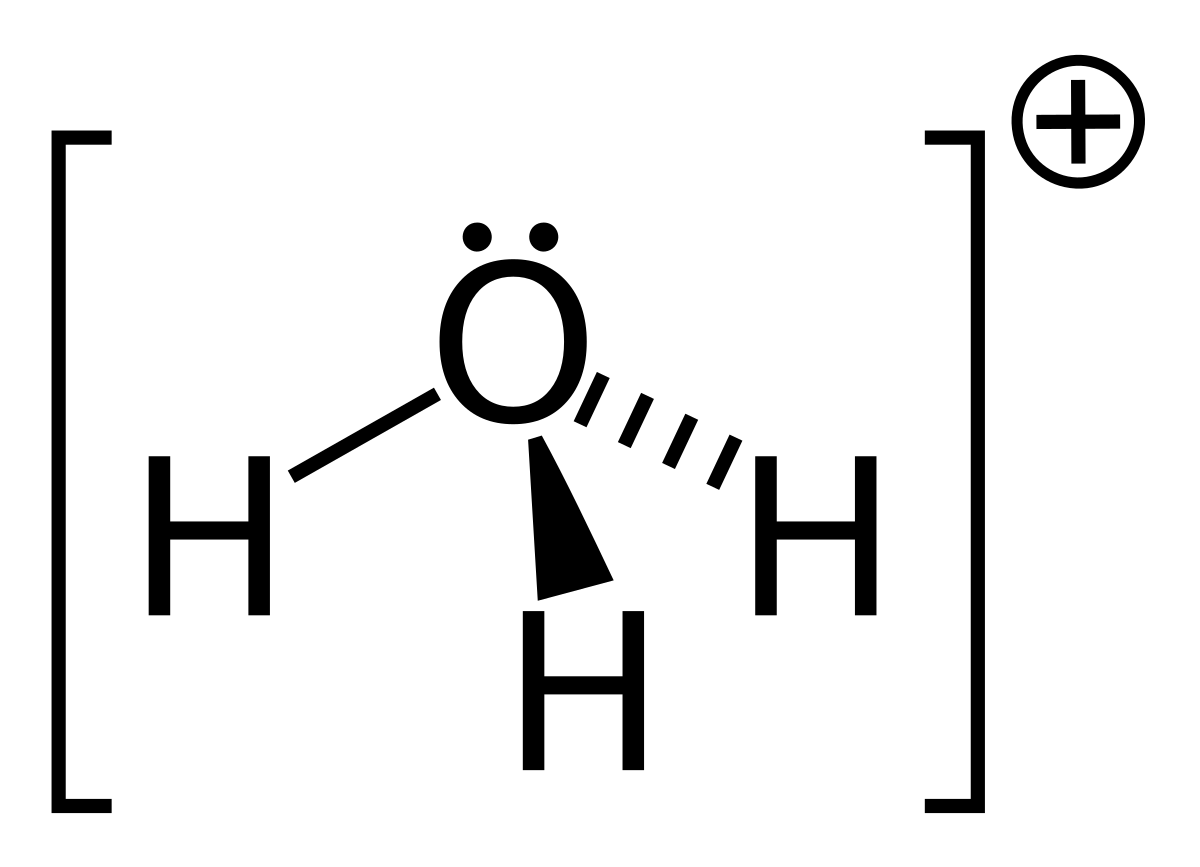


![Hydronium -[H3O]+ Hydronium -[H3O]+](http://www.chemtube3d.com/images/gallery/PNGfiles%20structures/I604ST01.png)



