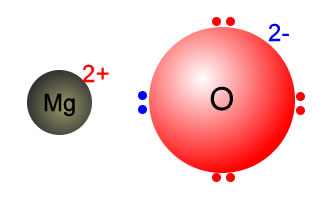
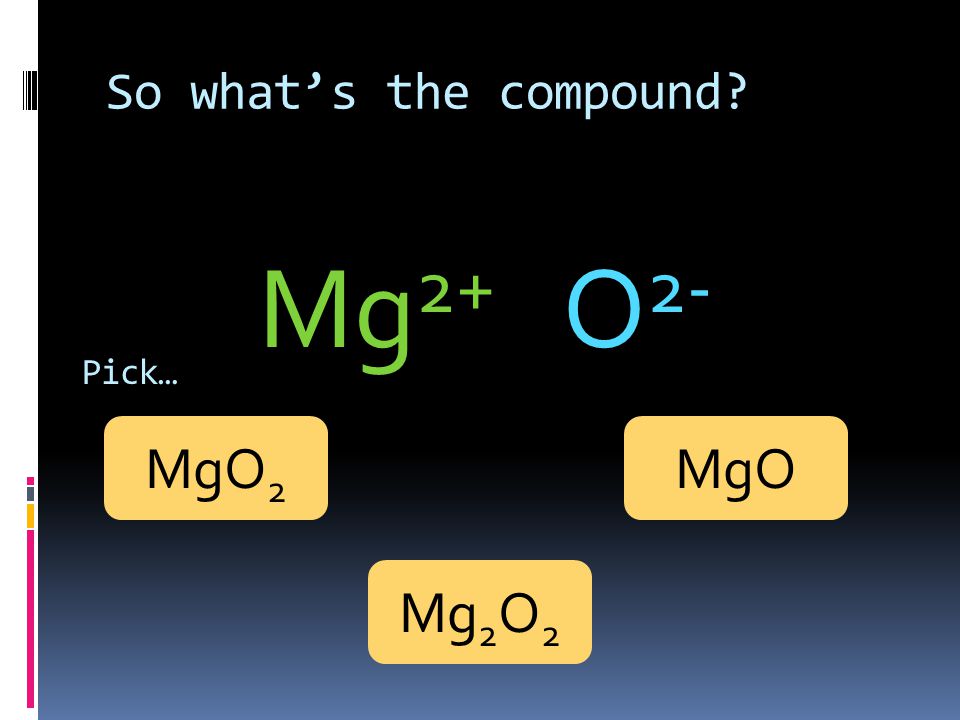
Why MgO exist as Mg^(2+)O^(2-) not as Mg^(o+)O^(ɵ) whereas the formation of Mg^(2+) from Mg requires more energy than formation of Mg^(o+) and formation of O^(ɵ) from O is exothermic whereas the

Why MgO exist as Mg^(2+)O^(2-) not as Mg^(o+)O^(ɵ) whereas the formation of Mg^(2+) from Mg requires more energy than formation of Mg^(o+) and formation of O^(ɵ) from O is exothermic whereas the
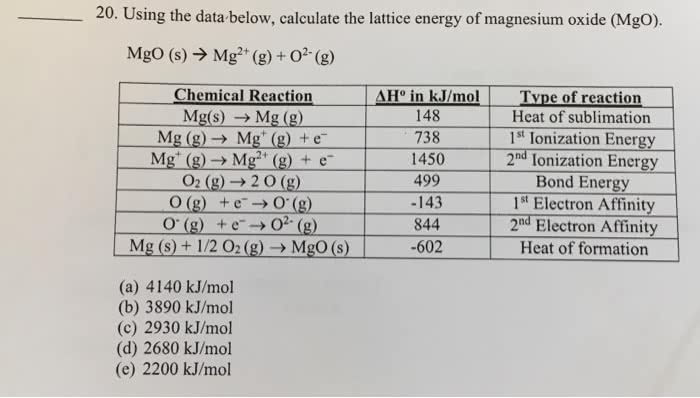
OneClass: The answer is B. Please show steps and explain. Thanks! 20. Using the data-below, calculate...

SOLVED: The formation reaction of magnesium oxide is as follows. Mg(s) + 0.5 O2(g) → MgO(s) This reaction can be constructed from the following reactions. In the blanks before each reaction, add