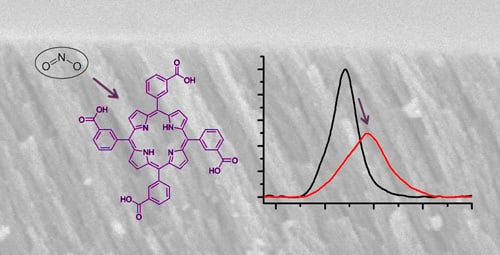
A robust approach to deriving long-term daily surface NO2 levels across China: Correction to substantial estimation bias in back-extrapolation - ScienceDirect

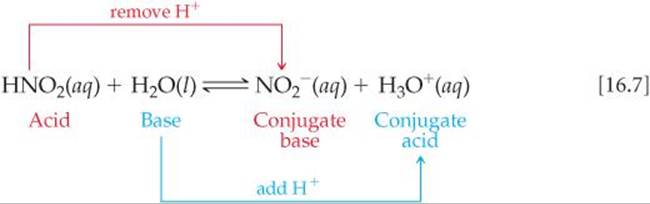
In this reaction, does NO2+ act like a Lewis acid, Lewis base, Bronsted acid or Bronsted base? A. Lewis Acid B. Lewis Base C. Bronsted Acid D. Bronsted Base E. Lewis Acid
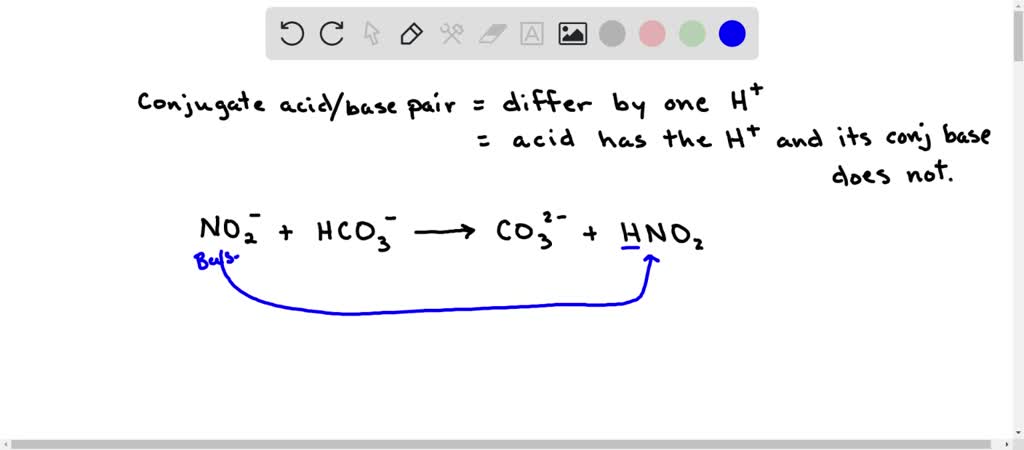
SOLVED: Consider the following reaction: NO2 - + HCO3 - ⇌ CO3 2- + HNO2 Identify the acid, base, conjugate acid and conjugate base.

1. What is the Bronsted Acid in the following equation: * NO2- +H2O HNO2 + OH- **a. NO2- **b. H2O **c. HNO2 **d. OH- 2. What is the Bronsted base in the
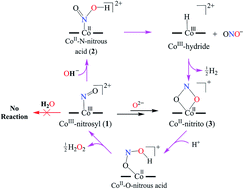
Nitric oxide monooxygenation (NOM) reaction of cobalt-nitrosyl {Co(NO)}8 to CoII-nitrito {CoII(NO2−)}: base induced hydrogen gas (H2) evolution - Chemical Science (RSC Publishing)